Integral membrane protein/amphipol complexes and high-resolution solution-state NMR spectroscopy.
6_ Applications: NMR studies of GPCRs associated to amphipols and of their ligands (2/2).
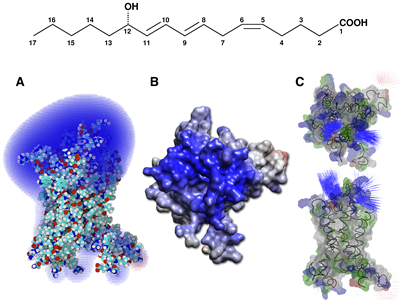
More recently, we proved experimentally and theoretically that NMR spectroscopy can be amenable to the study of high affinity ligand in their bound state (Catoire et al. (2011)). This is possible in the case of an inherent ultra-fast diffusive association of charged ligands onto a highly charged extracellular surface, and by slowing down the 1H–1H cross-relaxation by perdeuterating the receptor. The demonstration was based on the study of the heptadecanoid 12-HHT in interaction with the leukotriene BLT2 receptor associated to amphipol A8-35, in addition to the former study realized with LTB4. Indeed, 12-HHT exhibits an experimentally measured Kd value 62 nM. Kinetic measurements indicate that 12-HHT binds fast onto BLT2 (i.e. with an association rate constant kon of 2.9.108 M-1.s-1), exceeding by ~3 times the limit usually—but improperly—cited in the literature for biomolecular diffusional associations. This fast-associating kon is not physically unrealistic, however: even for large biomolecules, cases of protein-protein association have been reported with kon values close to or in excess of 109 M-1.s-1(Schreiber et al (1996),Gabdoulline et al (2002)).
In this case, electrostatic interactions prevail because of their long-range nature, while they do not affect koff, which is governed by short range interactions, including van der Waals and hydrophobic interactions, salt bridges and hydrogen bonds. Indeed, both agonists have a net charge of -1 and interact with the highly positively charged extracellular surface of the receptor.
The electrostatic potential of the latter was calculated for a model of the BLT2 receptor after a 0.5-μs molecular dynamics simulation in a fully hydrated lipid bilayer (Figure 13). This simulation suggests a readily accessible binding pocket located close to the surface (Figure 13), as observed for other class A GPCRs (Nygaard et al (2009)).
An animation of the electrostatic field around BLT2 was generated using SpiderMol (Callieri et al (2010))
providing an interactive view of proteins with animated particles and rendered surface.
The animation can be viewed on the following YouTube channel:
http://www.youtube.com/watch?v=1B6f_awCPL4 or http://www.youtube.com/marcbaaden
On the nuclear longitudinal relaxation timescale, this provides the opportunity to study the structure of tight-binding ligands, i.e. with Kd of a few tens of nM, bridging the gap between pharmacology and NMR.