AMPHIPOLS: Design, synthesis and properties of amphipol A8-35
One of the first APols to be designed, called A8-35 (Tribet at al.(1996)), has become by far the most extensively used APol. Its solution behavior has been studied in details by a broad range of physical techniques (Gohon et al. (2006, 2004), Tribet et al. (2009), Giusti et al. (2012)), and modeled by molecular dynamics (Perlmutter et al. (2011)). A8-35 is comprised of a relatively short polyacrylate chain (~35 acrylate residues), some of the carboxylates of which have been randomly grafted with octylamine (~9 of them) or isopropylamine (~14 units), for an average MW of ~4.3 kDa (Fig. 2). N.B. In earlier papers, the average length of A8 35 molecules had been estimated to ~70 residues, based on size exclusion chromatography (SEC) data using reference polymers as MW standards. Re-evaluation of this issue using direct methods (unpublished data by F. Giusti and J. Rieger) has led to the current value.
The ~12 acid groups that have remained free are charged in aqueous solutions (Gohon et al (2004)), which makes the polymer highly water-soluble (> 200 g.L-1), while the octylamide moieties render it amphipathic. Isopropylamide residues lower the charge density.
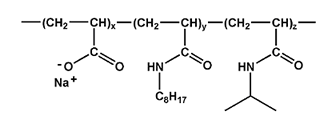
Figure 2. Chemical structure of amphipol A8-35; x ≈ 0.35, y ≈ 0.25, and z ≈ 0.4. From ref. Tribet et al (1996).
In aqueous solutions, A8-35 forms well-defined, small, globular particles (Gohon et al (2006)) above a critical association concentration of ~0.002 g.L-1 (Giusti et al. (2012)). Each A8-35 particle has a mass of ~40 kDa and, therefore, comprises an average of 9-10 A8 35 molecules (Gohon et al (2006)). MD simulations show the particles to be micelle-like, with a hydrophobic core and hydrophilic surface, and less fluid than detergent micelles (Perlmutter et al. (2011)).
Because the solubility of A8-35 depends on the presence of charges on its carboxylate groups, it is affected by every factor that will affect the latter, such as lowering the pH below neutrality or adding multivalent cations (Gohon et al (2006), Picard et al. (2006), Diab et al. (2007), Gohon et al. (2008)). This can create difficulties (due to aggregation) for some applications (see Table 1 in Applications), for which better-suited APols would be desirable (see Amphipols with different chemical structures). These limitations however seldom turn out to be a serious hindrance.
It has proven quite difficult and lengthy to develop, validate and characterize alternative chemical structures presenting as satisfying properties as those of A8-35. For this reason, most of the detailed studies of APols and MP/APol complexes to date have been carried out using this particular APol. This provides the putative user with a considerable body of information about what can and what cannot be attempted with it.
