AMPHIPOLS: Applications
An application that appears particularly promising is to use APols as a mild, stabilizing medium in which to fold membrane proteins (Pocanschi et al. (2006), Dahmane et al. (2011), Bazzacco et al. (2012)), including G protein-coupled receptors (Dahmane et al. (2009), Bazzacco et al. (2012),Banères et al. (2011)). The folding yields achieved for the latter are much higher than those observed in detergent or lipid/detergent solutions (Fig. 7). GPCRs folded in APols can then be, if need be, purified to near-homogeneity by affinity chromatography (Dahmane et al. (2009), Banères et al. (2011)).
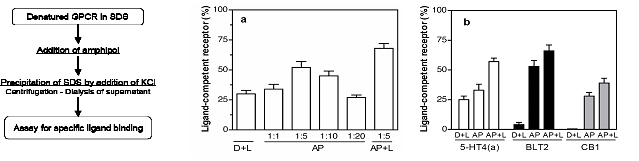
Figure 7. Amphipol-assisted folding of four G protein-coupled receptors. Folding in APol A8-35 (AP) or in a mixture of A8-35 and lipids (AP+L) is compared to folding in lipid/detergent mixtures (D+L). The receptors were obtained from inclusion bodies and solubilized in SDS under an inactive form. Folding in detergent was achieved by immobilizing on a column the SDS-solubilized receptors and substituting SDS with the detergent/lipid mixture (Banères et al. (2003)). Folding in APols was achieved by supplementing the receptors in SDS solution with either A8-35 or a 5:1 w/w A8 35/lipid mixture and removing the SDS by precipitation with KCl followed by dialysis (Pocanschi et al. (2006), Popot et al. (1987)). The extent of folding is given as the ratio between the number of binding sites for specific ligands and the number of receptor molecules initially present in the SDS solution, based on total protein content. (a) Folding of the leukotriene BLT1 receptor. The BLT1/A8-35 mass ratio was varied from 1:1 to 1:20, as indicated. (b) Folding of the serotonin 5 HT4(a), the leukotriene BLT2 and the cannabinoid CB1 receptors. Adapted from ref. (Dahmane et al. (2009)).
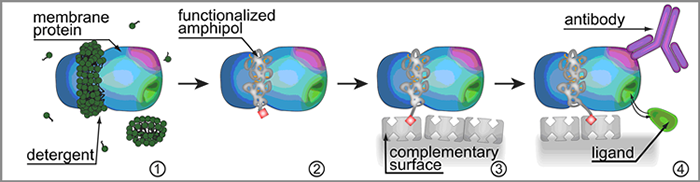
Another promising application is to use functionalized APols for immobilizing MPs onto solid supports (Charvolin et al. (2009), Pocanschi et al. (2006), Popot et al. (1987)). As long as APols are not displaced by another surfactant, their association with MPs, albeit non-covalent, is irreversible, and resists extensive washing with surfactant-free buffer (Popot et al. (2003),Tribet et al. (2009),Zoonens et al. (2007), Charvolin et al. (2009), Tribet et al. (1997)). Trapping a MP with a functionalized APol therefore results in a permanently functionalized complex (Zoonens et al. (2007)). This has innumerable possible applications. In particular, appropriately tagged APols can be used to immobilize the MPs they associate with onto solid supports in a simple, mild and versatile manner. The ligand-binding properties of the immobilized proteins can then be studied, in detergent-free solutions, by any convenient observation method, opening the way to a host of applications in diagnostics, drug discovery, or the search for natural biological partners ( Charvolin et al. (2009)) (Fig. 8).