Integral membrane protein/amphipol complexes and high-resolution solution-state NMR spectroscopy.
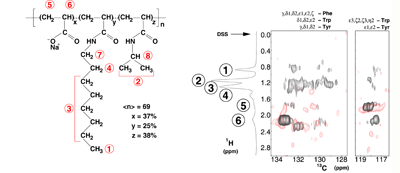
4_Organization of amphipols around membrane proteins.
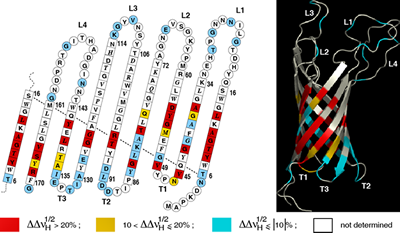
NMR spectroscopy has been used to examine the organization of membrane protein/A8-35 complexes, yielding an increasingly detailed view of protein-polymer interactions. A general description of the distribution of the alkyl chains was first obtained, taking advantage of the variation of dipolar environment experienced by the amide protons of tOmpA upon trapping with either A8–35 or its deuterated homolog (Zoonens et al. (2005)).
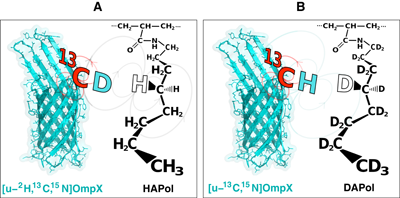
Two further studies exploited intermolecular 1H- 1H or 13C-1H dipole-to-dipole cross-relaxation phenomena (Figures 10). They yielded a qualitative detection of spatial proximities between 13C atoms in the side chain of aromatic residues of OmpX and 1H nuclei of A8–35 (Catoire et al (2009)) and the identification of hydrophobic contacts between specific amide protons of KpOmpA and octyl and isopropyl chains of A8–35 (M. Renault & A. Milon, unpublished data).
The three studies showed that contacts with the alkyl chains of A8–35 are confined to the transmembrane, hydrophobic surface of the proteins. This conclusion has been recently confirmed by direct visualization of the amphipol layer around a mitochondrial supercomplex by single particle cryo-electron microscopy (Althoff et al. (2011)). 1H/2H exchange measurements show that some of the amide protons of the membrane-spanning region of OmpX exchange much more readily than others, which likely reflects the dynamics of the barrel ( Catoire et al (2010b)).