Integral membrane protein/amphipol complexes and high-resolution solution-state NMR spectroscopy.
1_ Membrane proteins associated to amphipols are in their native fold.
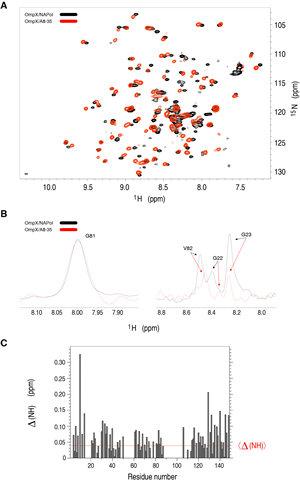
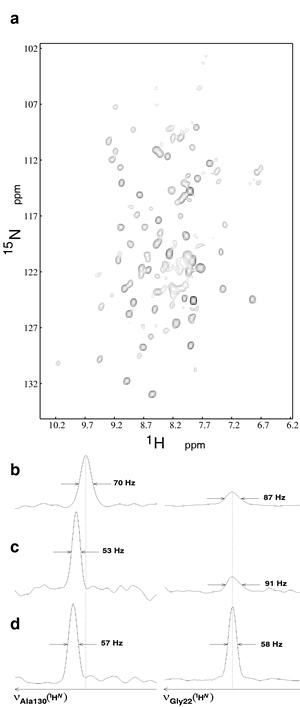
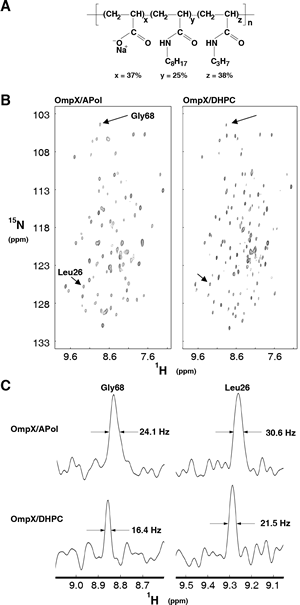
Early NMR experiments were aimed at better characterizing membrane protein/amphipol complexes and at exploring the resources and limitations of NMR to study their structure and dynamics. Among proteins chosen as models, two well-characterized β-barrel proteins from the outer membrane of E. coli (tOmpA and OmpX) were first tested in the laboratory. These proteins, whose 3D structures were initially solved by X-ray crystallography, have been extensively studied by solution NMR, mostly in the presence of detergent. NMR studies of their complexes with amphipols showed them to either retain or regain their native fold when associated to A8-35 ( Zoonens et al. (2005), Catoire et al (2010b)), to SAPols (Dahmane et al. (2011)), or to NAPols (Bazzacco et al. (2012)) (Figure 9).

![FIG9C:
Comparison of 2D [15N,1H]-TROSY spectra of SAPol-trapped [u-2H,15N]tOmpA acquired at 30°C (in black), 50°C (in red), and 70°C (in blue). Figure9: image 3 0f 4 thumb](../NMR_1/images_NMR_1/FIG1C_thumb.png)