
AMPHIPOLS: Amphipols with different chemical structures
In the initial stage of developing A8-35, it was shown that some changes could be brought to the chemical structure of the latter without compromising the ability of the resulting polymers to keep MPs soluble in the absence of detergents. This included longer polymers, with a MW in the 35 kDa range (A34-35, A34-75) (6), shorter ones (unpublished data), polymers with a higher density of charges (A8-75, A34-75) (6) or polymers carrying dodecyl rather than octyl hydrophobic chains (unpublished data).
Polymers similar to A8-35 but differing from it, mainly, by the distribution of the alkyl chains have also been proposed (27), as well as polymers that comprise zwitterionic groups and carry no net charge at basic pH (but, because the anionic group is a carboxylate, positive charges in acidic solutions) (22).
Similarly, phosphorylcholine-based APols owe their high solubility to their globally neutral but highly charged polar heads, as well as to the presence of a secondary amine function (11, 13, 31).
Sulfonated APols are being developed with the view of mimicking A8-35's properties while ensuring solubility at acidic pH, mainly for NMR purposes (10, 32). The search for totally non-ionic APols has been a protracted endeavor, due to the difficulty to make these molecules soluble enough without driving the formation of huge particles and complexes, which would be of no use for most applications. The feasibility of the concept was established relatively early, using the hydroxyl moieties of Tris groups to confer hydrophilicity (33), but the solubility of this family of molecules turned out to be too difficult to control for convenient use. More recently, very promising results have been obtained with glycosylated non-ionic APols (34, 35).
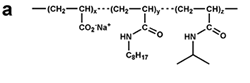
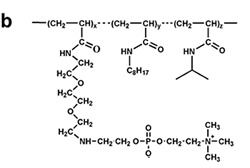
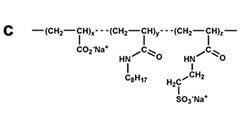
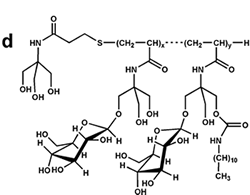
Figure 7. Chemical structure of some amphipols. (a) A8-35, the reference, polyacrylate-based APol; see refs. 6-8. (b) C22-43, a phosphorylcholine-based APol; see refs. 11, 13, 31. (c) A sulfonated APol; see refs. 10, 32. (d) A glucosylated, non-ionic APol; see refs. 34, 35. From ref. 5.
In most cases, the ability of these new APols to keep MPs soluble in the absence of detergents has been demonstrated, usually by simple centrifugation experiments. Few studies have been reported of the size and physical and biochemical stability of the complexes thus produced (see e.g. refs. 10, 11, 32, 35). Some of these new molecules may become important tools, once their use has been validated, for specific applications where A8-35 is suboptimal, such as MP solution NMR studies (32) or crystallization, or can not be used at all, such as isoelectrofocusing. Parallel studies comparing the properties of various families of APols and their ability to stabilize panels of MPs (see e.g. refs. 10, 11, 32, 35) are, of course, extremely desirable in order to enable potential users to pick their choice.