AMPHIPOLS: Applications
Overview
-
Folding and Immobilization of Membrane Proteins
The original aim behind the design of APols was to make it possible to handle and study in aqueous solutions MPs that are destabilized or whose functional properties are perturbed by detergents. While stabilization indeed seems fairly general, it may be acquired at the cost of making some experimental approaches more difficult, as is the case for NMR solution studies and, most conspicuously, for crystallization. Before embarking upon a given study, one has, therefore, to weigh the pros and the cons of resorting to APols. A schematic overview of those applications of APols, proven or hypothetical, for which there exist published or unpublished data is given in Table 1 (updated from ref. 4). For more details, see ref.5.
| Type of application | Rationale | Observations | References |
| Stabilization | A complex issue. Includes limitation of hydrophobic sink, preservation of MP/lipid interactions, and damping of transmembrane helix movements | Most MPs tested to date are more stable in APols than in detergent solutions. See protein trapping | (1, 6, 9, 10, 18, 20) |
| Functional studies | Avoid perturbations and/or destabilization by detergents. | Ligand binding very generally unperturbed. No perturbation of BR or nAChR functions. Slowing down of calcium ATPase enzymatic cycle, probably due to damping of large scale transmembrane conformational changes. | (1, 9, 10, 16, 21, 22) |
| Folding/refolding | The mildness of APols, along with other factors, makes them an excellent environment to fold or refold denatured MPs, such as those produced as inclusion bodies. | Published data concern two porins, BR, and four GPCRs. | (17, 18) |
| Electron microscopy | APols ought to be particularly useful to study fragile MPs and MP supercomplexes. | Better spread of particles in cryo-EM. A particularly promising application that still has to be fully explored. | (9, 25, 26) |
| MP immobilization onto solid supports | Properly functionalized APols will simultaneously make a MP water-soluble, stabilize it biochemically, and anchor it onto a solid support. | Hitherto applied to detecting antibody binding by surface plasmon resonance (on chips) and binding of a neurotoxin by fluorescence microscopy (on beads). | (16) |
| Delivery of MPs to preexisting membranes | APols do no lyse target membranes (lipid vesicles or black films, cell plasma membrane) and can therefore be used to deliver to them hydrophobic cargoes such as MPs. | MPs have been delivered both to lipid vesicles and to black films. Caveats: insertion process expected to be traumatic for fragile MPs; carrier APols will remain associated to the target membrane. | (17, 27) |
| NMR | Usual conditions for solution NMR of MPs are highly aggressive (high detergent concentrations, high temperature); APols may permit to study MPs that do not stand them. A8-35 is easier to deuteriate than most detergents. | MP/APol particles are slightly larger than the smallest MP/detergent complexes, which lowers the resolution. The greatest current limitation to date has been that A8-35 aggregates at pH<7. Full-fledged application of APols to solution NMR awaits the coming of age of pH-insensitive APols. | (14, 19, 28, 32, 36) |
| Mass spectrometry | Analysis of APol-trapped MPs | Doable. Study in progress. | (19) |
| Isoelectrofocusing | Improving yields over that in detergent solutions? | Requires strictly neutral APols. Study in progress. | |
| Trapping labile supercomplexes, assembly intermediates etc. | Observing and studying MP assemblies that are too labile to resist exposure to detergents. | Perhaps one of the most interesting applications of APols, but still underdeveloped. | |
| Cell-free synthesis | Letting MPs synthesized in vitro fold in a mild environment. | Under study. | |
| X-ray crystallography | Stabilizing MPs under crystallization conditions. | Chemical structure of A8-35 far from ideal (charges). Current data limited to low resolution diffraction by tertiary MP/APol/detergent complexes. | (15) |
| Applications of fluorescent APols | Those go from following the distribution of APols and studying their interaction with MPs or with target membranes or cells to FRET experiments. | Used to study the irreversibility of MP/APol association and the displacement of MP-bound APols by unlabeled APols or detergents, the critical association concentration of APols etc. | (12, 13) |
Table 1. A schematic overview of various validated or foreseeable applications of APols to MP studies and their current state of development. The second column gives examples of the rationales for using APols for the application considered, the third one a very brief summary of current observations and, if applicable, caveats.
Two recently developed applications appear particularly promising. One of them is to use APols as a mild, stabilizing medium in which to fold membrane proteins (17), including G protein-coupled receptors (18). The folding yields achieved for the latter are much higher than those observed in detergent or lipid/detergent solutions (Fig. 5).
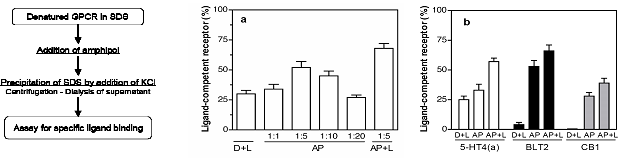
Figure 5. Amphipol-assisted folding of four G protein-coupled receptors. Folding in amphipol A8-35 (AP) or in a mixture of A8-35 and lipids (AP + L) is compared to folding in lipid/detergent mixtures (D + L). The receptors were obtained from inclusion bodies and solubilized in SDS under an inactive form. Folding in detergent was achieved by immobilizing on an affinity colum the SDS-solubilized receptors and substituting SDS with the detergent/lipid mixture (29). Folding in amphipols was achieved by supplementing the receptors in SDS solution with either A8-35 or a 5:1 w/w A8-35/lipid mixture and removing the SDS by precipitation with KCl followed by dialysis (17, 30). The extent of folding is given as the ratio between the number of binding sites for specific ligands and the number of receptor molecules initially present in the SDS solution, based on total protein content. (a) Folding of the leukotriene BLT1 receptor. The BLT1/A8-35 mass ration was varied from 1:1 to 1:20, as indicated. (b) Folding of the serotonin 5 HT4(a), the leukotriene BLT2 and the cannabinoid CB1 receptors. Adapted from ref. 18.
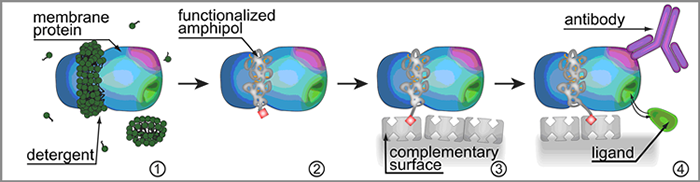
Another interesting application is to use functionalized APols for immobilizing MPs onto solid supports (16). As long as APols are not displaced by another surfactant, their association with MPs, albeit non-covalent, is irreversible, and resists extensive washing with surfactant-free buffer (1, 12, 13, 16, 24). Trapping a MP with a functionalized APol therefore results in a permanently functionalized complex (12). This has innumerable possible applications. In particular, appropriately tagged APols can be used to immobilize the MPs they associate with onto solid supports, in a particularly mild and versatile manner. The ligand-binding properties of the immobilized proteins can then be studied, in detergent-free solutions, by any convenient observation method, opening the way to a host of applications in diagnostics, drug discovery, or the search for natural biological partners (16) (Fig. 6).
For a discussion of the relative advantages and drawbacks of amphipols, fluorinated surfactants and nanodiscs as alternatives to detergents to handle membrane proteins in aqueous solutions, see ref. 5.