Membrane Protein Biochemistry Lab
Biochimie des Protéines Membranaires
Our laboratory gathers biologists, physicists and chemists who are interested in the structure, structural dynamics and physical chemistry of membrane proteins, either in membrane mimetic systems (liposomes or nanodiscs), or in classical detergent solutions or alternative surfactants such as amphipols.
Following the footsteps of Jean Perrin who created the institute together with Baron Edmond de Rothschild, we develop fundamental, interdisciplinary research for health and environment.
The team
Latest News
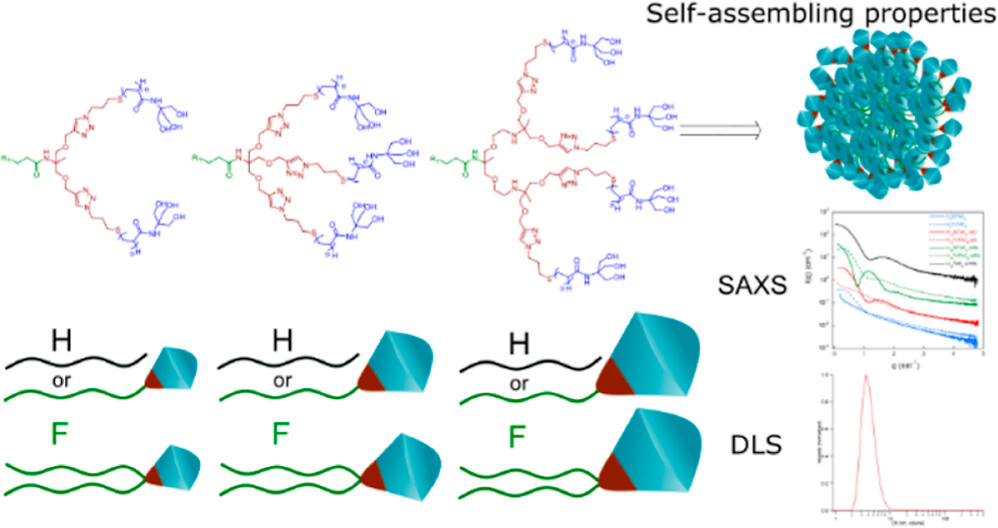
Macromolecules Publication
Congratulations to Françoise who co-published in Macromolecules ! Synthesis, Characterization,

20240422_membrane digest
A bitter sweet symphonie (that’s life) of super cool membrane-related papers !

20240415_membrane digest
From toxin translocation to cryotomo of MDa complexes, the membrane’s digest is once again multi scale !
Seminars
Facts and Figures
Research is a collective adventure where cohesion of the team is essential. We foster social activities to stimulate exchange of expertise, mentoring and synergies between research topics. Social life around the Institute is highly attractive for students and early researchers.
in the lab
PhD defense
Trained students
Publications
Collaborations